
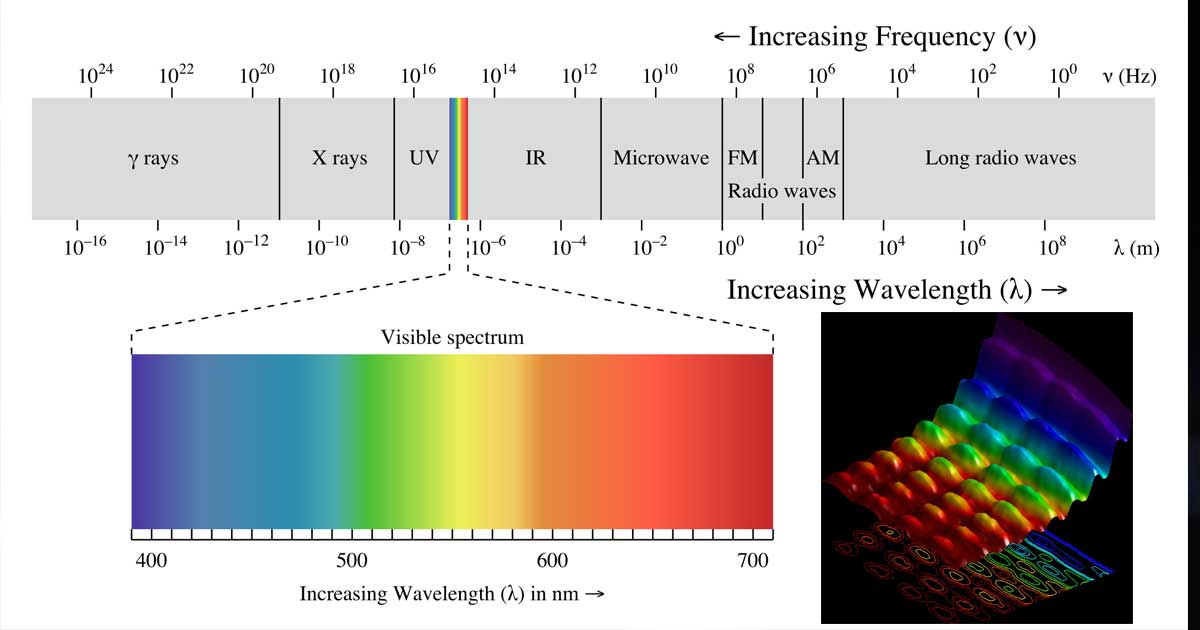
That is, if we see an emission spectrum anywhere that looks exactly like this one, then we know that we’re looking at an emission spectrum of helium. What this means is that if we take this helium spectrum, for example, then we know that any helium gas anywhere in the world will produce this exact same emission spectrum with the exact same emission lines at the exact same wavelengths. The important thing to realize is that an emission spectrum kind of acts like a fingerprint for a particular element. Now, since we can see that there’s a whole load of lines in this emission spectrum, we’re not going to individually read off the wavelength of every single one. Meanwhile, the wavelength of this line is about 565 nanometers. So, for example, we can see that this emission line here from the spectrum of the unknown gas has a wavelength of about 454 nanometers. 
At the top of this figure, we’ve got a wavelength scale, which allows us to read off the wavelength of each of these emission lines. If we look at our unknown gas spectrum, we can see a load of these bright lines, which are emission lines of this particular gas. What we need to do then is to identify a match between this emission spectrum of the unknown gas and one of these five known emission spectra. But rather it’s entirely one of these five elements whose emission spectra were shown here. That is, it’s not a mixture of some number of different elements. Since we’re asked which one of these five elements the unknown gas is, this means we know that the unknown gas must be a pure sample. We’re being asked to identify which of these five elements is the unknown gas. Then, below this, we’ve got the emission spectra of helium, oxygen, neon, argon, and xenon, each of which is a pure gaseous element. Up at the top, we’ve got the spectrum of the unknown gas that the scientist wants to identify. Taking a look at this figure that we’ve been given, we can see that we’ve got a series of emission spectra. Which of the five elements is the unknown gas? Also shown in the figure are the emission spectra of five pure gaseous elements. In order to identify the gas, he looks at the spectrum of visible light emitted from it when it is heated. Department of Energy, by the National Aeronautics and Space Administration, by NIST's Standard Reference Data Program (SRDP), and by NIST's SMA Program.A scientist has a sample of an unknown gas. This database was funded by the Office of Fusion Energy Sciences of the U.S. Online: March 1995 - Last update: October 2022 All rights reserved. NIST reserves the right to charge for these data in the future. Department of Commerce on behalf of the United States. Past Contributors: Haris Kunari, Jean E. Sansonetti, Jeffrey R. Fuhr, Larissa I. Podobedova, Wolfgang L. Wiese, John J. Curry, Gerry R. Dalton, Robert Dragoset, Fun-Chen (Jesse) Jou, William C. Martin, Peter J. Mohr, Arlene Musgrove, Craig J. Sansonetti, and Gloria Wiersma Students contributing to data entry: Eric Carpentier, Thomas Carpentier, Amy Zimmerman, Adrian Hamins-Puertolas, Marko Hamins-Puertolas, Anna Sharova, Genevieve Tan NIST ASD Team Principal Developers (Currently Active): Alexander Kramida, Yuri Ralchenko, and Joseph Readerĭata Compilers (Currently Active): Alexander Kramida, Edward B. Salomanĭatabase Developers (Currently Active): Alexander Kramida, Yuri Ralchenko, and Karen Olsen The Data Center is located in the Physical Measurement Laboratory at the National Institute of Standards and Technology (NIST). The Atomic Spectroscopy Data Center has carried out these critical compilations. This database provides access and search capability for NIST critically evaluated data on atomic energy levels, wavelengths, and transition probabilities that are reasonably up-to-date. Help - On-line help in using the database.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
